Pharmaceutical research
In contrast to other mistletoe products, the standardised manufacture of abnobaVISCUM is performed by using a patented pressing procedure (EP 0 288 603 A1). Using this manufacturing process to produce the pressed juice ensures that a high yield of mistletoe lectins and viscotoxings and the entire range of plant ingredients is transferred to the extract. The plant extract contains 75% of the plant material used. With a buffer solution, the various concentrations of abnobaVISCUM are then produced.
Plant material from both a summer and a winter harvest is used. These two raw materials are processed separately to pressed juice and are subsequently mixed in another patented procedure (EP 0 310 984 B1). This mixture is produced because investigations have shown that various pharmacologically active substances are present in the plant to different extents, depending on the season. For example, particularly large quantities of polysaccharides are found in the white mistletoe berries, which are only harvested in winter.
The particular form of extracting abnobaVISCUM affects the formation of liposomes in abnobaVISCUM. Liposomes are cell-like structures in or to which one can include or attach active substances. Liposomes can thus enable targeted transport of the active substances to the “site of action”, i.e. the tumour. The medical term for this process is “drug targeting”.
The picture below shows you how liposomes with a size of approx. 100 nanometers look like.

Detail of an electron microscopic image of liposomes in abnobaVISCUM.
Liposomes contained in abnobaVISCUM are among the subjects of our current research, which characterises the liposomes and investigates their impact on the efficacy and tolerability of the medicinal product. Moreover, new applications for high accumulation and release of mistletoe compounds in the tumour tissue are prepared. New liposome production technologies have been established and international patents are currently filed (PCT/EP2019/0132 and PCT/EP2019/0133). Innovative liposomal formulations of mistletoe will soon be investigated pre-clinically.
Pharmaceutical research is generously recognised by public funding, for example:
Central Innovation Programme (SME) (small and medium-sized enterprises)
(Zentrales Innovationsprogramm Mittelstand (ZIM))
http://www.zim-bmwi.de
European Union: Marie Curie Actions
http://ec.europa.eu/research/mariecurieactions/index_en.htm
Together with the Universities of Utrecht (NL), Uppsala (S) and Karlsruhe (D).
Examples:
Marie Curie Actions in collaboration with the Universities Utrecht, Uppsala and Karlsruhe
“Novel drug deliver system produced by centrifugal technologies – composed to minimize adverse immune reactions and designed for optimized therapeutic effects.”
http://www.decentaid.eu/
One of the results of this research is that the “mode of action” of ABNOBA products and the destruction of cancer cells by lectins can be explained in detail. Cf. the following picture and its explanation:
How lectins initiate the programmed cell death of cancer cells, referred to as apoptosis:
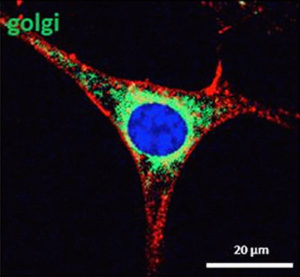
The picture shows the cell nucleus (blue) inside the cancer cell which is already penetrated by mistletoe lectins (red) that reached the cytosol. By contacting the ribosomes which they inactivate they induce he programmed cell death. The Golgi apparatus is stained in green. Only ABNOBA mistletoe preparations provide this scientific evidence of the active principle (the scale bar is 20 µm; 1 micrometre [µm] is equivalent to 1/1,000th of a millimetre).
You can also follow how the cancer cells are ”destroyed“ in a time-lapsed film composed of images made by confocal microscopy. Click on the following links:
Video 1
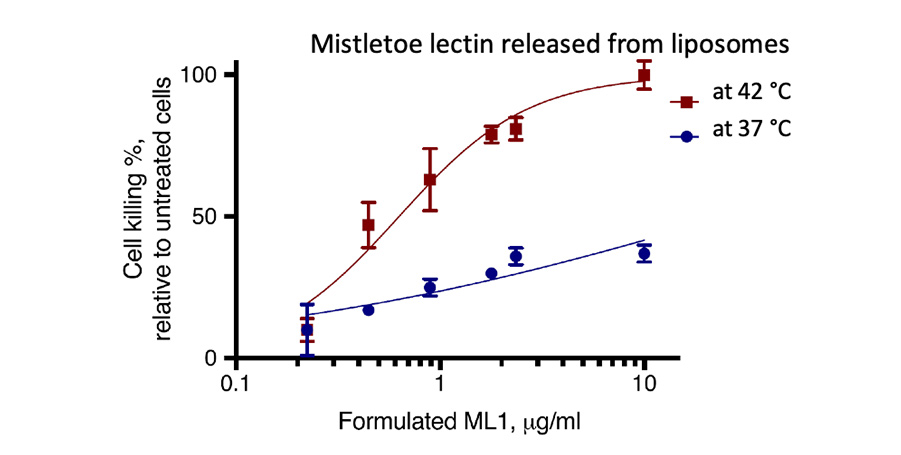
Another result of the European collaboration is the release of mistletoe lectins from liposomes, induced by local hyperthermia and their cytotoxicity against murine colon carcinoma cells (CT26): The figure below shows that at 42°C mistletoe lectins are fully released from liposomes and show apoptotic activity like non-encapsulated mistletoe lectin with an half-inhibitory concentration (IC50) of 0,6 μg/ml. In contrast, mistletoe lectins are only marginally released at 37°C with an IC50 of 25 μg/ml.
Literature source of results and figure:
https://www.sciencedirect.com/science/article/pii/S0939641118305915?via%3Dihub
University hospital of Freiburg:
Uni-Zentrum für Naturheilkunde
Viscum Album neutralizes tumor-induced immunosuppression
in a human in vitro cell model